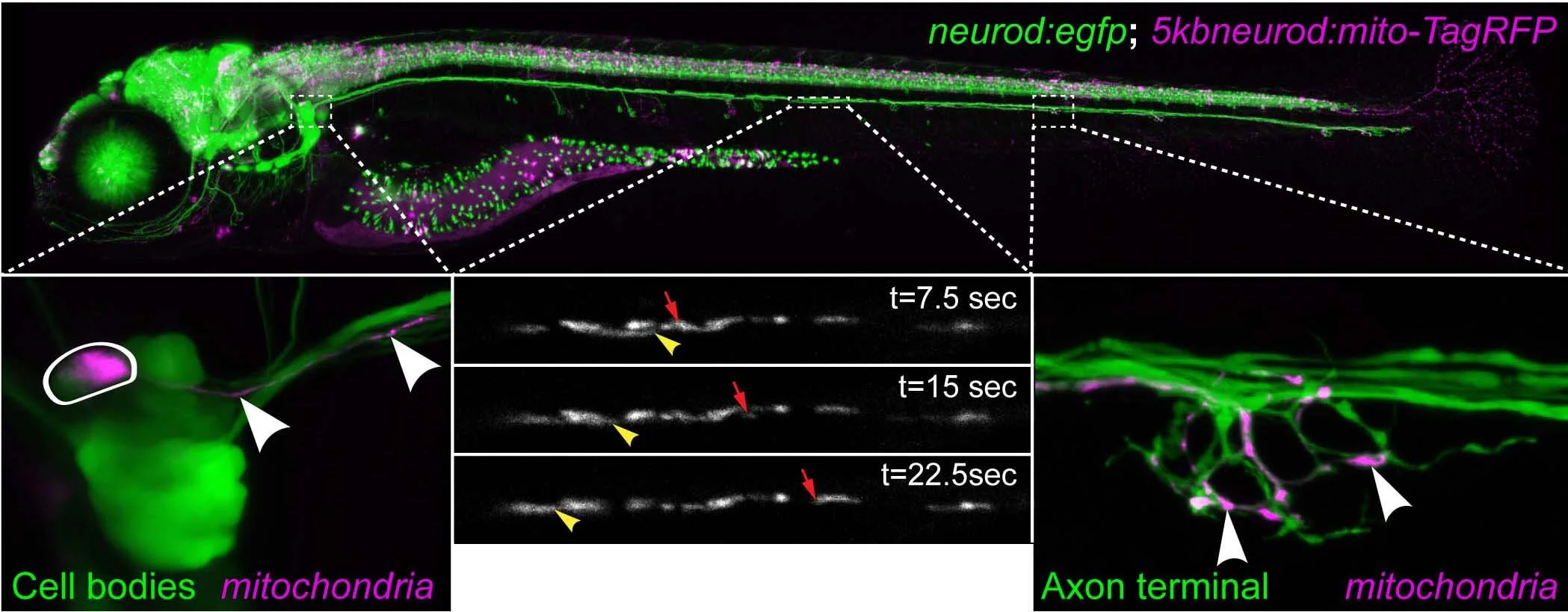
We use zebrafish neurons and mammalian cells to study the cell biology of neural circuit formation and function. Zebrafish offer a number of advantages for this work including: 1) optical transparency; 2) genetic accessibility; 3) hardiness; and 4) fast development. Contact us to learn more!
Current Projects
Mitochondrial transport: mechanisms and function
Mitochondria are actively transported through neuronal processes called axons. This “axonal” transport moves mitochondria either toward microtubule plus ends (anterograde - pointed away from the cell body) or microtubule minus ends (retrograde - pointed towards the cell body). How and why these organelles move is of high interest for many reasons, including the correlation between abnormal mitochondrial positioning and neurodegenerative diseases. We have identified novel regulators of mitochondrial retrograde transport and are pursuing questions involving their molecular mechanisms and the functional ramifications of disrupting this process.
In vivo analysis of Dynein regulation
A diverse array of cargo are moved by microtubule motors in axons. This movement must be highly regulated in order for proteins and organelles to be correctly localized for use and recycling. In a simplistic model, anterograde OR retrograde motors could be selectively attached to these cargos for movement. However, a growing amount of evidence suggests that some cargos, including late endosomes, autophagosomes, and mitochondria, bind to both anterograde and retrograde motors simultaneously. From this work, it has been determined that regulatory mechanisms must be in place to turn one motor “on” and one “off” for processive movement. These mechanisms are likely also cargo-specific. We are interested in identifying these molecular regulators of directional transport for cargos in axons.
Genetic screen for modulators of retrograde axonal transport
While we have learned much about the biophysics of dynein movement, how this single motor protein complex attaches to an undefined number of cargos is still a subject of intense investigation. To compliment this work, our lab utilizes forward genetics in zebrafish to identify novel regulators of dynein-mediated cargo movement. To date, this screen has yielded novel insights into lysosome and mitochondrial attachment to the dynein motor. We have many uncharacterized mutants and are still actively screening to identify more of the proteins that can modulate dynein-cargo attachment and retrograde transport.